Metal halide perovskites refer to a large family of crystalline materials with structures similar to calcium titanate. This natural mineral was named after Russian mineralogist Lev Perovski.
Two-dimensional metal halide perovskite materials possess excellent optoelectronic properties such as strong light absorption, high fluorescence quantum yield (PLQY), and versatile chemical processability. They demonstrate great promise towards practical applications such as solar cells, LEDs, lasers, and photocatalysts.
Due to the presence of lead and heavy halides, two-dimensional perovskites systems have large spin-orbit coupling (SOC), which in the presence of structural distortions could lift the spin-degeneracy near the conduction and valance band-edges, adding the potential of optospintronic applications.
Optospintronic applications connect the spin degrees of freedom to light and charge. It could lower power consumption and achieve faster switching times compared with conventional electronic systems. To realize such an optospintronic device for two-dimensional perovskite, the spin properties of the materials need a better understanding.
Assistant Professor Xihan Chen and his team from the Department of Mechanical and Energy Engineering at the Southern University of Science and Technology (SUSTech) have recently made progress in the spin polarization lifetime of two-dimensional perovskites.
Their research, entitled “Tuning Spin-Polarized Lifetime in Two-Dimensional Metal–Halide Perovskite through Exciton Binding Energy,” was published in the Journal of the American Chemical Society (JACS), one of the most prestigious and top journals in chemistry.

Using ligand engineering to tune spin polarization lifetime of two-dimensional perovskites
Two-dimensional perovskites usually have a Ruddlesden-Popper structure. That is, the inorganic layer of PbI6 octahedrons are connected through the top corners through organic ammonium cations (see Figure 1a-b).
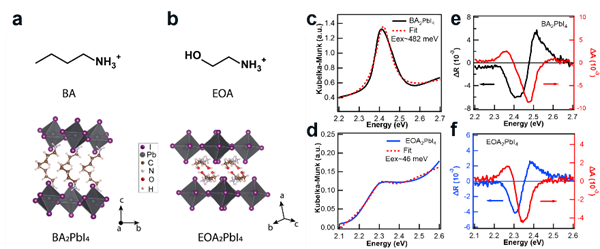
Figure 1. Structure of BA and EOA based two-dimensional perovskites and the corresponding transient reflection spectra.
The mismatch of the dielectric constant between the organic and inorganic layers causes the excitons generated by light absorption to be localized in the inorganic layer. As a result, the exciton binding energy is usually on the order of hundreds of millielectron volts. For example, the exciton binding energy of BA2PBI4 with a non-polar ligand (BA, butylamine) is 482 meV.
When using ethanolamine (EOA) ligands with a large dielectric constant (εr=37.2), the exciton binding energy can be reduced to 46 meV. The lower binding energy can effectively screen electronic interaction from the lattice, thereby increasing the spin polarization lifetime of the carriers. Therefore, the use of ligand engineering to tune the spin polarization lifetime of two-dimensional perovskites is promising for studying the physical mechanism of spin relaxation of such low-dimensional semiconducting materials.
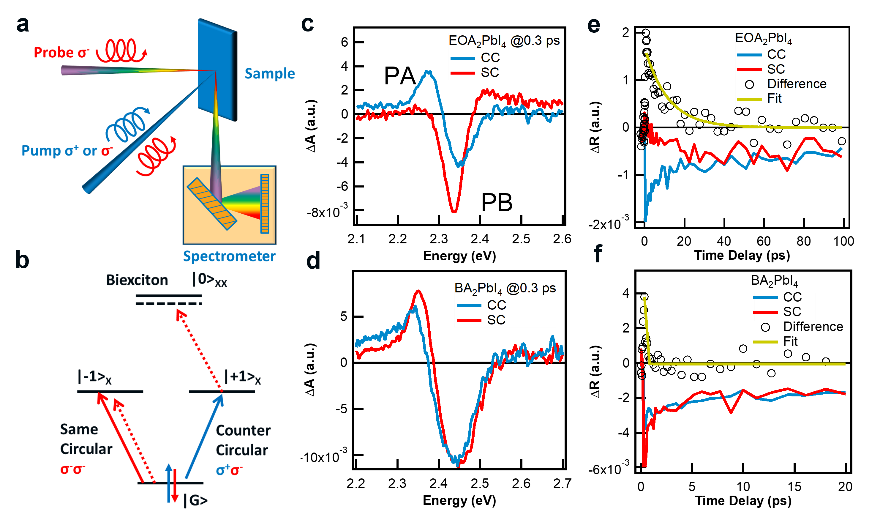
Figure 2. Schematics of transient circular polarized spectroscopy and the corresponding spin-polarization relaxation kinetics.
Based on this strategy, Prof. Chen’s team and Dr. Matthew C. Beard from the National Renewable Energy Laboratory (NREL) developed a series of two-dimensional perovskite single crystals with different organic ligands (Figure 1). Ultrafast spectroscopy is used to study the spin polarization lifetime and relaxation mechanism of photogenerated excitons in two-dimensional perovskite single crystals.
As shown in Figure 1, the increase in the dielectric constant of the ligand leads to a significant decrease in the binding energy of excitons. The transient reflection signal was transformed by the Kramer-Kronig method to obtain the corresponding transient absorption spectrum. The shape and position of the spectrum show that the generation of the bleaching signal mainly comes from the filling of free carriers or the state filling caused by the occupation of excitons.
Since the bleaching signal contains the common contribution of free carriers and excitons, a biexciton signal is used to represent the spin polarization and relaxation process. The ultrafast spectroscopic method is shown in Figure 2a-2b. The ultrafast circular polarization spectroscopy found that the EOA spin polarization lifetime was significantly enhanced compared with BA, reaching 26 ps (Figure 2). By changing the temperature and the carrier concentration, it was found that the temperature had a relatively small effect on spin relaxation. Therefore, the increase of the carrier concentration would reduce the spin polarization lifetime (Figure 3).
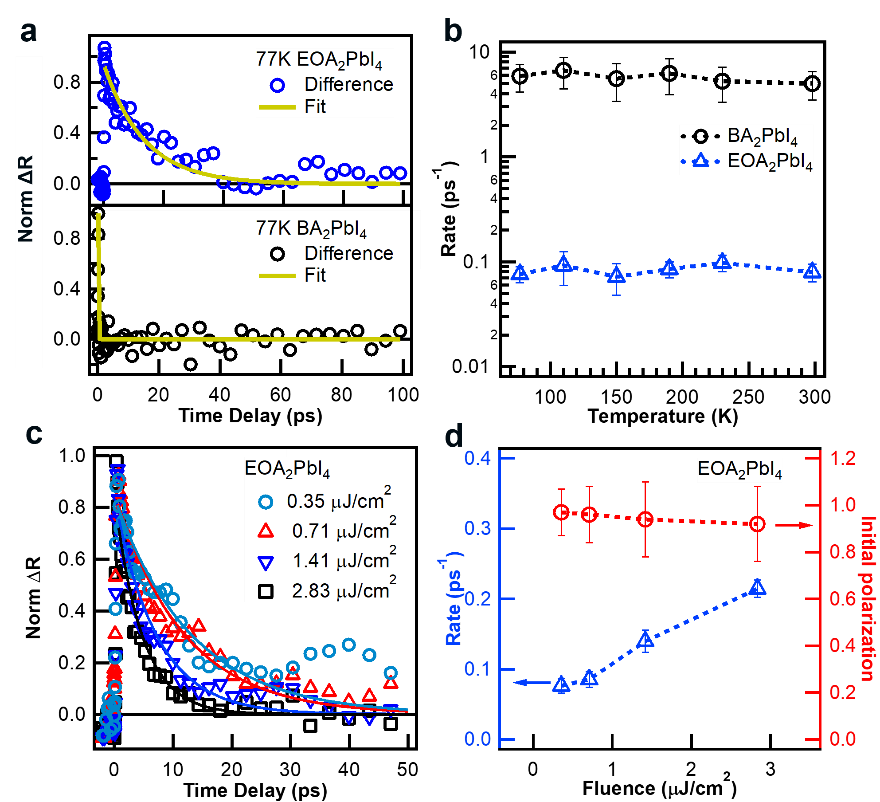
Figure 3. Temperature and carrier density dependence on spin-polarization lifetime.
Through the above research, it is inferred that at low concentrations, the D’yakonov-Perel (DP) mechanism plays a leading role in spin relaxation. In contrast, at high exciton concentration, Bir-Aronov-Pikus (BAP) mechanism plays a major role. Such that tuning exciton binding energy could tune spin-polarization lifetime. (Figure 4).
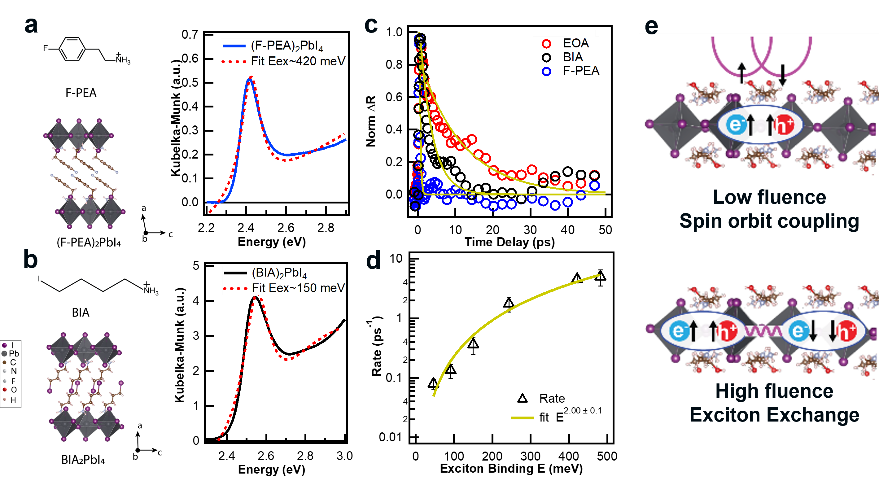
Figure 4. Spin-relaxation mechanism in two-dimensional perovskites, at low fluence, D’yakonov-Perel (DP) mechanism plays a leading role in spin relaxation. In contrast, at high exciton concentration, Bir-Aronov-Pikus (BAP) mechanism plays a major role.
Assistant Professor Xihan Chen from SUSTech is the first and corresponding author of this paper. Dr. Matthew C. Beard is also the corresponding author. Assistant Professor Haipeng Lu from the Hong Kong University of Science and Technology (HKUST) and Dr. Kang Wang from Xiamen University are the co-first authors.
This research was supported in part by the National Natural Science Foundation of China (NSFC).
Paper link: https://pubs.acs.org/doi/10.1021/jacs.1c08514
To read all stories about SUSTech science, subscribe to the monthly SUSTech Newsletter.
Proofread ByAdrian Cremin, Yingying XIA
Photo By