Electric vehicles are very limited in mileage due to the low energy density of batteries. Developing batteries with high energy density and long calendar life is urgently needed. Commercial lithium (Li)-ion batteries are already very close to their ceiling values. Further substantial increase in energy density depends on developing new electrode materials.

Associate Professor Meng Gu’s research group from the Southern University of Science and Technology (SUSTech) recently revealed the atomic nanocomposite structure and 3D structure of LiMetal and SEI using low-dose cryogenic high-resolution transmission electron microscopy (Cryo-HRTEM) and cryogenic electron tomography (Cryo-ET).
Their study, entitled “Cryo-Electron Tomography of Highly Deformable and Adherent Solid-Electrolyte Interphase Exoskeleton in Li-Metal Batteries with Ether-based Electrolyte”, was published in Advanced Materials, a scientific journal covering materials science.
Compared with commercial graphite anode (~372 mAh/g), Li metal with the body-centered cubic structure (LiBCC) is the ultimate anode with the highest specific capacity (~3860 mAh/g), which could attribute to higher energy density. However, research shows that lithium metal’s (LiMetal) ultra-high chemical and electrochemical reaction activity would lead to the corrosion of electrolyte to LiMetal, which further brings about the growth of lithium dendrite and a series of problems. Regulating electrolytes and additives can tune the morphology and component of the SEI and suppress the growth of dendrite. However, because SEI and LiMetal are sensitive to air, moisture, and electron beam, their atomic structure can hardly be detected, not to mention the 3D structure.
With ether-based DOLDME-LiTFSI electrolyte, LiF and Li2O nanocrystals are formed and embedded in a thin but tough amorphous polymer in the polymer-rich surficial passivation layer (SEI exoskeleton). The fast Li strips along [110] or [121], which produces eight exposed {111} planes at halfway charging (Figure 1).
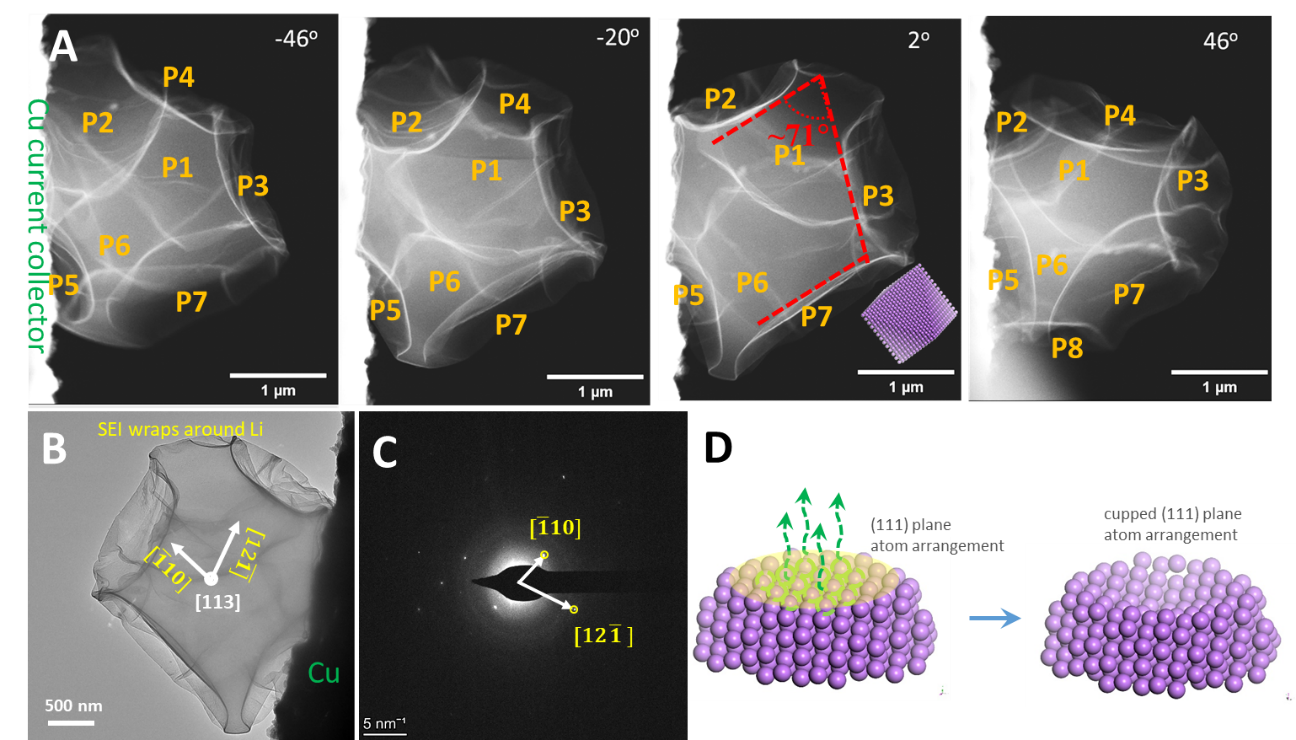
Figure 1. (A) Cryo-STEM tomography tilt series viewing the morphology. (B) TEM image of a halfway stripped Li particle. (C) Corresponding [113] zone electron diffraction pattern of the particle in panel B. (D) Atomic model showing the cupped surface induced by atom stripping along the (111) plane.
After full Li stripping, completely sagging and empty SEI husks are formed, which can sustain extensive bending and buckling, with the smallest bending radius of curvature observed approaching tens of nanometers without apparent damage (Figure 2).
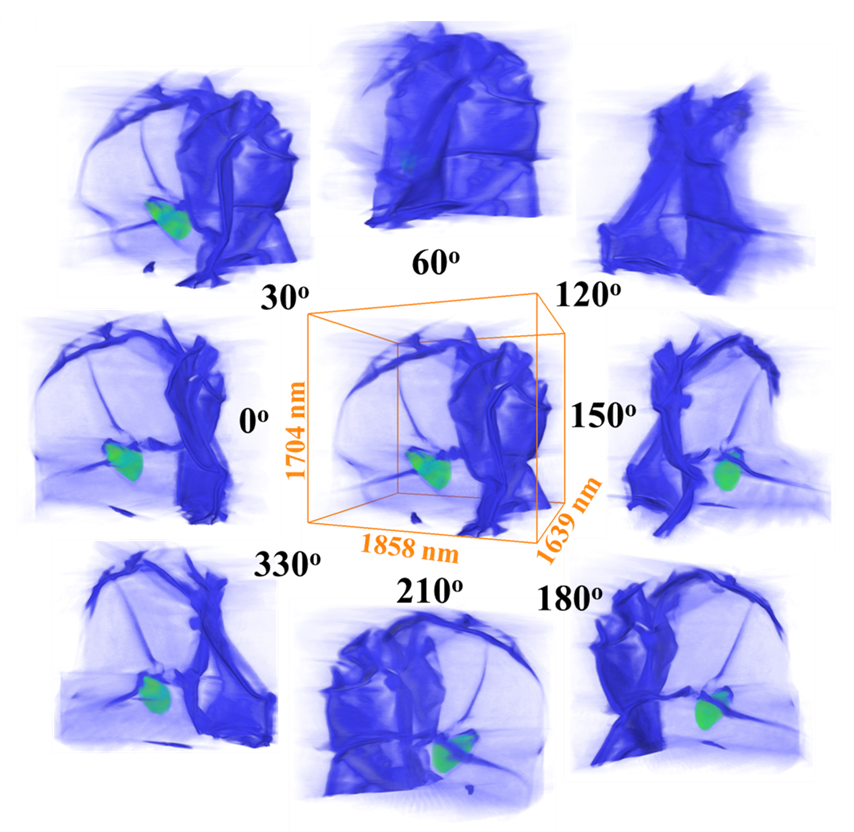
Figure 2. Morphology of SEI and residual Li after full stripping
In the second round of Li plating, a thin LiBCC sheet firstly nucleates at the current collector, extends to the top end of the deflated SEI husk, and then expands its thickness. The apparent zero wetting angle between LiBCC and the SEI interior means that the heterogeneous nucleation energy barrier is zero. Due to the stable chemo-mechanical properties and complete-wetting property of SEI to Li, the SEI can essentially prevent further reactions between LiMetal and electrolyte, which explains the superior performance of Li-metal batteries with ether-based electrolytes. However, uneven refilling of the SEI husks results in dendrite protrusions and some new SEI formation during the second plating. A strategy to form bigger SEI capsules during the initial cycle with higher energy density than following cycles enables further enhanced Coulombic efficiency to above 99%.
Dr. Xiangyan Li from Prof. Meng Gu’s research group at SUSTech and Dr. Bing Han from the University of California, San Diego (UC San Diego) are the first authors of this paper. Prof. Meng Gu and Ju Li from the Massachusetts Institute of Technology (MIT) are the corresponding authors. SUSTech is the first affiliation of the paper.
This work was supported by the Guangdong Innovative and Entrepreneurial Research Team Program, Guangdong-HongKong-Macao Joint Laboratory for Photonic-Thermal-Electrical Energy Materials and Devices, and the Shenzhen Science and Technology Program. The authors also acknowledge Dr. Peiyi Wang and Dr. Xiaomin Ma at the Cryo-EM Center of SUSTech for their assistance on the microscope operation.
Paper link: https://doi.org/10.1002/adma.202108252
To read all stories about SUSTech science, subscribe to the monthly SUSTech Newsletter.
Proofread ByAdrian Cremin, Yingying XIA
Photo By