Chemistry is the foundation of scientific research. It is related to all aspects of people’s living, and is essential to the development of society. For example, the discovery of artemisinin has saved the lives of a large number of malaria patients. A large number of chemical reactions are needed to discover drugs, particularly in the pharmaceutical industry.
The Ugi four-component reaction (Ugi-4CR) is widely used in drug discovery. This reaction was discovered in the 1960s and played a pivotal role in organic chemistry. However, the molecules obtained by conventional Ugi-4CRs are racemates (a pair of enantiomers when the product possesses a stereocenter) that are challenging to separate. Also, it is very difficult and inefficient to obtain only one of the isomers by existing asymmetric synthetic means. For half a century, scientists around the world have not found an effective way to synthesize only one enantiomer of Ugi-4CR.

On September 14, Professor Tan Bin from the Department of Chemistry at Southern University of Science and Technology (SUSTech), published an online article entitled “Asymmetric phosphoric acid-catalyzed four-component Ugi reaction” in the international academic journal Science. His breakthrough in basic research has solved the scientific problem that synthetic chemists have been challenging for nearly 60 years, namely how to achieve effective stereocontrol of Ugi-4CR. Professor Tan Bin, the corresponding author of the study, is the first recipient of the National Science Fund for Distinguished Young Scholars program trained by SUSTech. Another corresponding is Professor K. N. Houk from the University of California, Los Angeles. Zhang Jian, a doctoral student of Tan Bin’s research group, and Yu Peiyuan, a postdoctoral fellow of Professor K. N. Houk, are co-first authors.
The research was funded by the National Natural Science Foundation of China, the SUSTech President Fund, the Shenzhen Science and Technology Commission. This achievement was SUSTech’s first Science article published as the first communication unit with complement of all experimental process at SUSTech, which witnessed the rapid development of the research strength of SUSTech.
The Ugi-4CR assembles peptide-like α-acylaminoamides through one-pot reaction of a carbonyl compound, an amine, an acid, and an isocyanide. Ugi-4CR is well suited for diversity-oriented synthesis applicable in drug discovery, as it facilitates rapid access to diverse libraries of biologically important molecules. The high step economy and atom efficiency of the reaction, as well as its convergent nature, foster its wide use in the synthesis of heterocyclic scaffolds, natural products, macrocycles, polymers, and other target molecules. Despite these practical advantages, the long-standing stereochemical challenges of the Ugi reaction have yet to be fully addressed. Consequently, access to chiral Ugi products for drug candidate exploration is hindered.
The chiral phosphoric acid (CPA) framework was targeted as a catalyst for asymmetric Ugi-4CR. The heightened acidity of CPAs over carboxylic acids is perceived to accelerate the kinetics of the enantioselective Ugi reaction so as to outcompete the background reaction. Also, self-assembled heterodimerization between the CPA and carboxylic acid brings about a dual effect: enhanced acidity of the catalyst and nucleophilicity of the carboxylic acid. Both of these favor the catalytic enantioselective Ugi-4CR. A myriad of well-established or custom CPAs with well-defined chiral pockets could be readily applied, potentially leading to complete stereocontrol. A CPA that could suppress the Passerini and other side reactions would enable rapid imine formation and its preferential activation over the carbonyl group.
A catalytic asymmetric Ugi-4CR was accomplished with 1,1′-spirobiindane-7,7′-diol (SPINOL)–derived CPA4 and CPA6 as organocatalysts. The reaction exhibited broad substrate compatibility and good to excellent enantioselectivity [up to 99% enantiomeric excess (ee)]. Activation of the imine might be accomplished by CPA–carboxylic acid heterodimer catalysis via a bifunctional activation mode, which was supported by experiments (carboxylic acids with varying pKa values and steric properties yielded products with a range of ee values) and density functional theory (DFT) calculations (lowest energy among all the considered activation modes). The calculated free energy profile for the catalytic Ugi reaction gave three CPA-combined key transition states, which highlighted the bifunctional property of the CPA. In the favored enantio-determining transition states, the aryl groups fit into the pocket formed by the two substituents (cyclohexyl rings) of the catalyst, revealing the importance of noncovalent interactions in controlling the stereochemical outcome of this reaction.
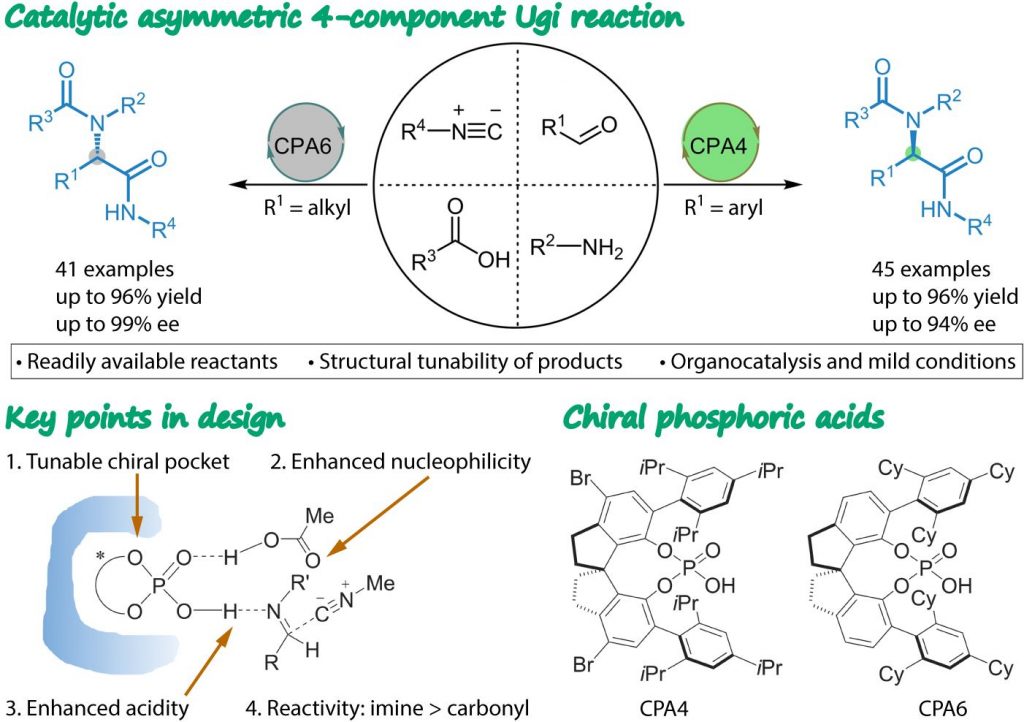
This operationally simple one-pot enantioselective Ugi-4CR harnesses inherent benefits of multicomponent reaction and organocatalysis to access up to 86 enantioenriched α-acylaminoamides, which are otherwise challenging to obtain via conventional methods, from four achiral building blocks in excellent yields and enantioselectivities. DFT calculations gave a detailed catalytic mechanism, especially with respect to activation modes and enantio-determining transition states. Because amide functionality constitutes the defining primary linkage in proteins, we foresee multiple uses of this asymmetric four-component Ugi protocol for the synthesis of chiral peptides and components of natural products. We also anticipate that this work will initiate the further development of asymmetric multicomponent chemistry.
Article link: http://science.sciencemag.org/content/361/6407/eaas8707
Proofread ByXia Yingying
Photo ByDepartment of Chemistry