N6-methyladenosine (m6A), the most widely distributed modification in mRNA, could mediate the processes of mRNA splicing, export, degradation, and translation. The deficiency of m6A has been shown to result in serious defects in the development and functions of the nervous system. However, how and which m6A readers mediate these functions remains to be elucidated.
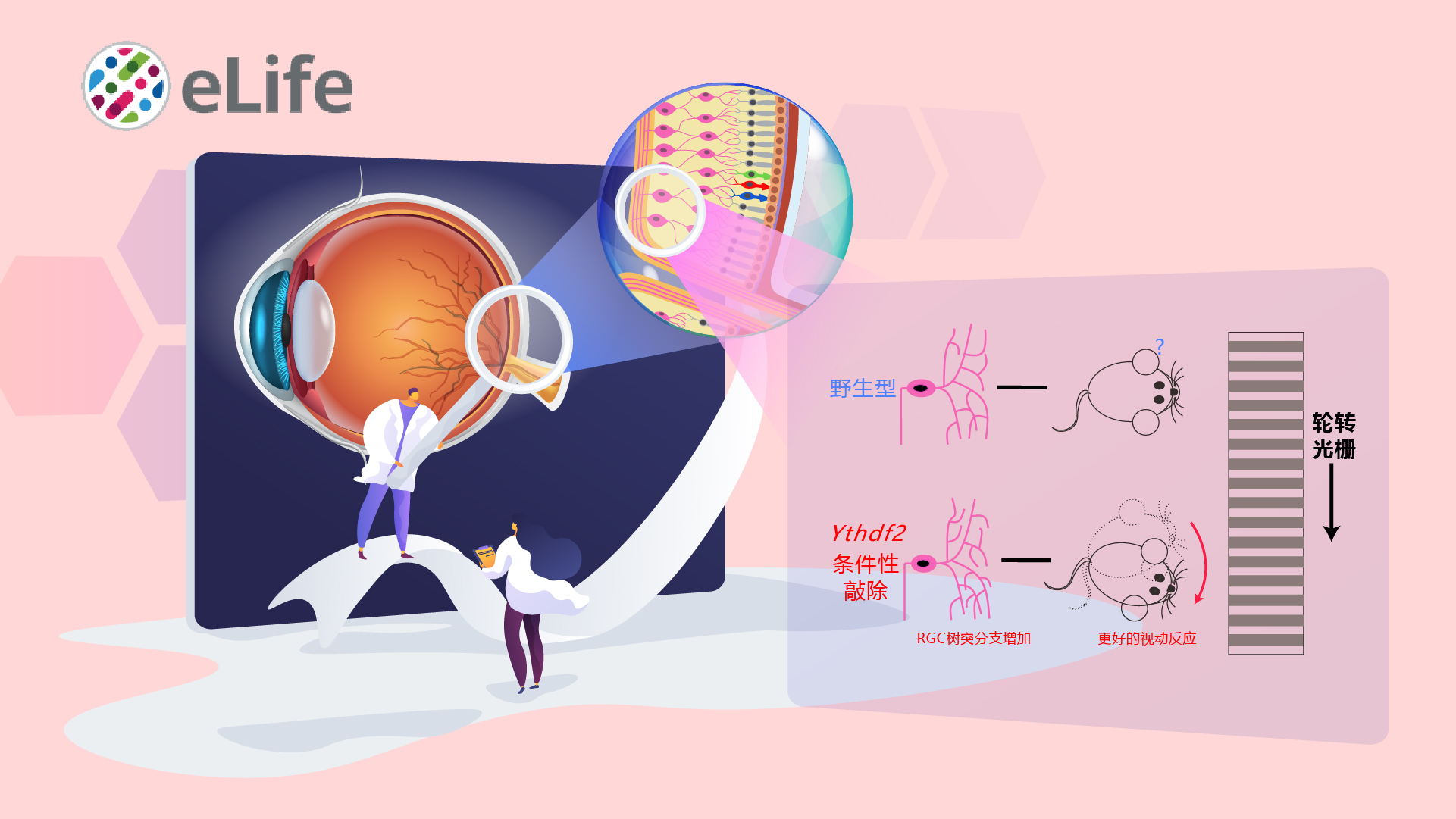
Prof. Shengjian Ji’s group from the School of Life Sciences at Southern University of Science and Technology (SUSTech) recently published a research article entitled “The m6A reader YTHDF2 is a negative regulator for dendrite development and maintenance of retinal ganglion cells” in eLife. Their study elucidates that m6A reader protein YTHDF2 plays an important role in regulating dendrite development and functions of retinal ganglion cells (RGCs) and also influences the neurodegeneration progress of RGCs.

Figure 1. Online publication of the paper in eLife
The previous works from Prof. Shengjian Ji’s group showed that m6A modification plays an essential role in the early development of the nervous system, especially in the process of axon development (Yu et al., Nucleic Acids Research, 2018; Zhuang et al., Nucleic Acids Research, 2019; Yu et al., Advanced Science, 2021). They found that the m6A eraser FTO could regulate axon growth by affecting the local translation of mRNA in the axon. They further reported m6A readers YTHDF1 and YTHDF2 could synergistically control axon growth of cerebellar granule cells under physiological conditions by regulating local translation of key molecules in the Wnt/PCP signaling pathway. However, whether m6A modification regulates retinal nervous system development is still unknown.
This recent research found that the expression level of m6A reader YTHDF2 was high in RGCs, and knockout or knockdown of YTHDF2 led to the increase of RGC dendritic arbors. Conditional knockout of Ythdf2 in the developing mouse retina increased the thickness and synapses numbers of the retinal inner plexiform layer. Furthermore, they discovered the better visual acuity of Ythdf2 condition knockout (cKO) mice compared with control in the optomotor assay. By utilizing multi-omics analysis, they identified Kalrn, Strn, Ubr4 as targeted mRNAs by YTHDF2 and further confirmed that the regulation of RGC dendrite development by YTHDF2 is mediated by those targeted mRNA. Interestingly, they further found that the expression level of YTHDF2 was significantly increased in a glaucomatous model and verified Hspa12a and Islr2 as targeted mRNAs regulated by YTHDF2 in mediating the maintenance of RGCs in the neurodegeneration progress.
In summary, this data suggests that m6A reader YTHDF2 could regulate the processes of RGC dendrite development and maintenance in neurodegeneration by two groups of targeted mRNAs, respectively.
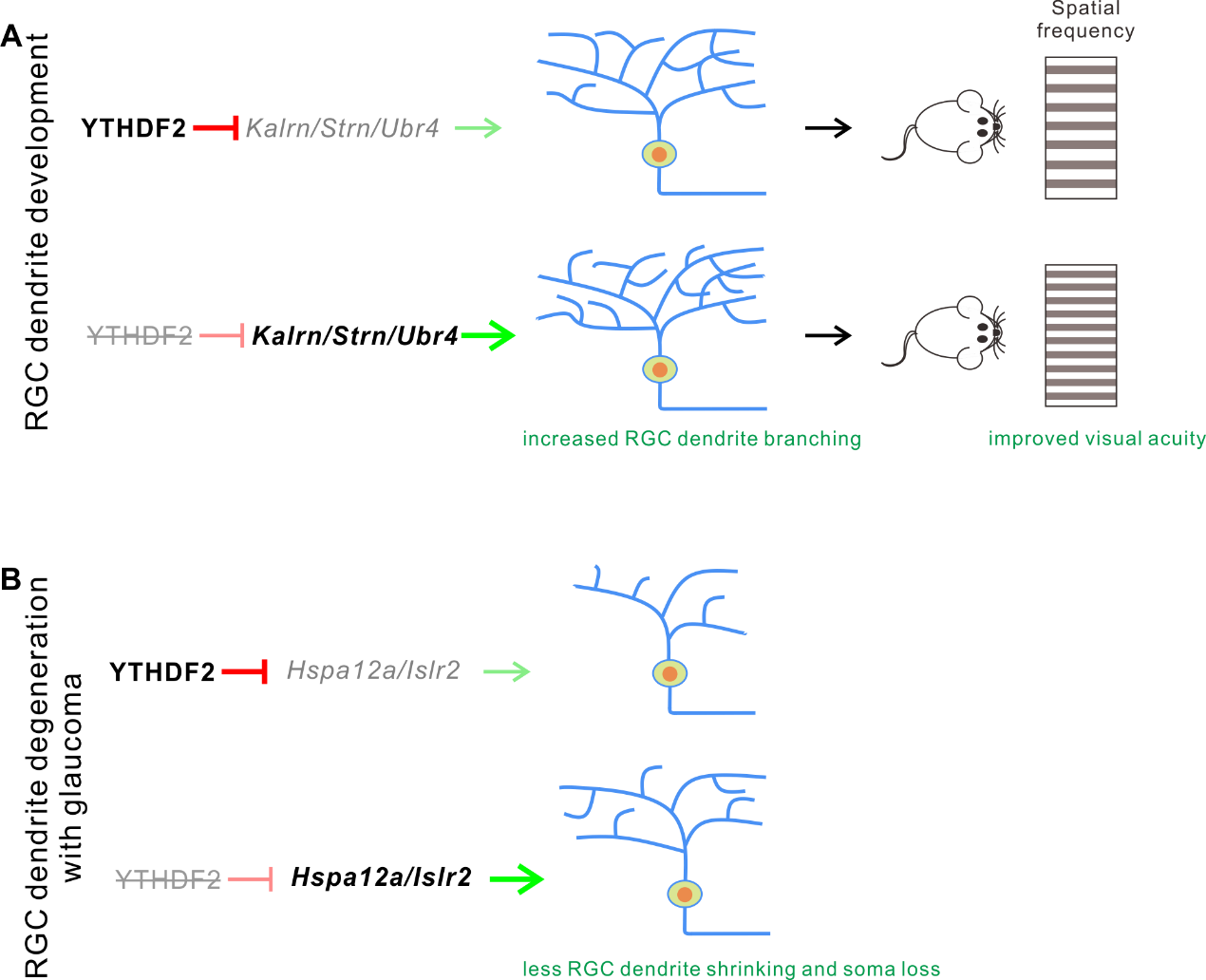
Figure 2. Working models for YTHDF2 in RGC dendrite development and maintenance
Fugui Niu, a doctoral student of the Joint Graduate Program at SUSTech-HIT (Harbin Institute of Technology), is the first author of this paper. Prof. Shengjian Ji at SUSTech and Prof. Bo Peng at Fudan University are the co-corresponding authors.
Paper link: https://elifesciences.org/articles/75827
To read all stories about SUSTech science, subscribe to the monthly SUSTech Newsletter.
Proofread ByAdrian Cremin, Yingying XIA
Photo By