Thermoelectric materials can directly convert heat into electricity and are widely used in precision temperature control, deep-space exploration, and industrial waste heat recovery. However, their energy conversion efficiency has long been limited by an incomplete understanding of how microscopic structures determine macroscopic transport properties. Recently, a research team led by Chair Professor Jiaqing HE from the Department of Physics at Southern University of Science and Technology (SUSTech) uncovered intrinsic chemical inhomogeneity in bismuth telluride–based thermoelectric materials at the atomic scale. The study systematically clarifies its formation mechanism and its decisive role in thermoelectric transport. By establishing a quantitative relationship between bonding polarity, electronic band gap, and the Seebeck coefficient, the researchers further develop a framework linking atomic structure to macroscopic thermoelectric performance, enabling direct prediction of macroscopic properties from atomic-scale elemental distributions. This work, titled “Atomic-Scale Chemical Inhomogeneity as a Determinant of Thermoelectric Transport in Bi2Te3-Based Materials,” was published in the flagship physics journal Physical Review X.

Bi2Te3-based materials are among the most efficient thermoelectric systems operating near room temperature and are widely used in micro-refrigeration and high-precision temperature control devices. Alloying with antimony (Sb) or selenium (Se) has long been an effective strategy for improving their performance. It has long been assumed that in the Bi2Te3-xSex solid solution, Se atoms first fully occupy the central Te(2) sites within the quintuple-layer structure before entering the outer Te(1) sites. However, using aberration-corrected scanning transmission electron microscopy combined with a newly developed data-denoising method, the SUSTech team directly revealed the atomic distribution of Se in the quintuple layers (Fig. 1). Their results show that Se atoms simultaneously occupy both Te(1) and Te(2) sites across all compositions, but consistently exhibit strong enrichment in the central layer, forming pronounced local chemical inhomogeneity. This behavior is observed throughout the entire composition range, indicating that it is an intrinsic structural characteristic of the Bi2Te3-xSex system rather than the uniform distribution assumed in ideal solid-solution models. Through atomic-scale compositional quantification, the researchers determined the exact Se occupancy at each layer and constructed an accurate atomic model incorporating this local inhomogeneity, revising previous structural models based solely on averaged crystallographic information.

Figure 1. Local chemical inhomogeneity of Bi2Te3-xSex.
To understand the origin of this local chemical inhomogeneity, the researchers combined atomic-scale charge density imaging with electron energy-loss spectroscopy. These measurements revealed intrinsic bonding differences between the central and outer layers in Bi2Te3 (Fig. 2). The outer-layer Te atoms exhibit stronger negative charge characteristics, whereas the central-layer Te atoms show distinct charge distributions and chemical shifts, indicating that the Bi–Te bond in the central layer is more covalent while the outer-layer bond is more ionic. This finding provides the first direct atomic-scale experimental evidence for a theoretical prediction proposed more than sixty years ago regarding bonding disparity in Bi2Te3. Owing to these layer-dependent differences in bonding energy and electronic character, the more electronegative Se atoms preferentially substitute the central Te(2) sites during alloying, thereby lowering the formation energy and stabilizing the observed chemical inhomogeneity. First-principles calculations combined with thermodynamic modeling further show that the spatial distribution of Se atoms is governed by the competition between formation enthalpy and configurational entropy. The experimentally observed distribution corresponds to thermodynamic equilibrium at the synthesis temperature, indicating that the chemical inhomogeneity in Bi2Te3-xSex arises from the combined effect of bonding disparity and finite-temperature thermodynamics.
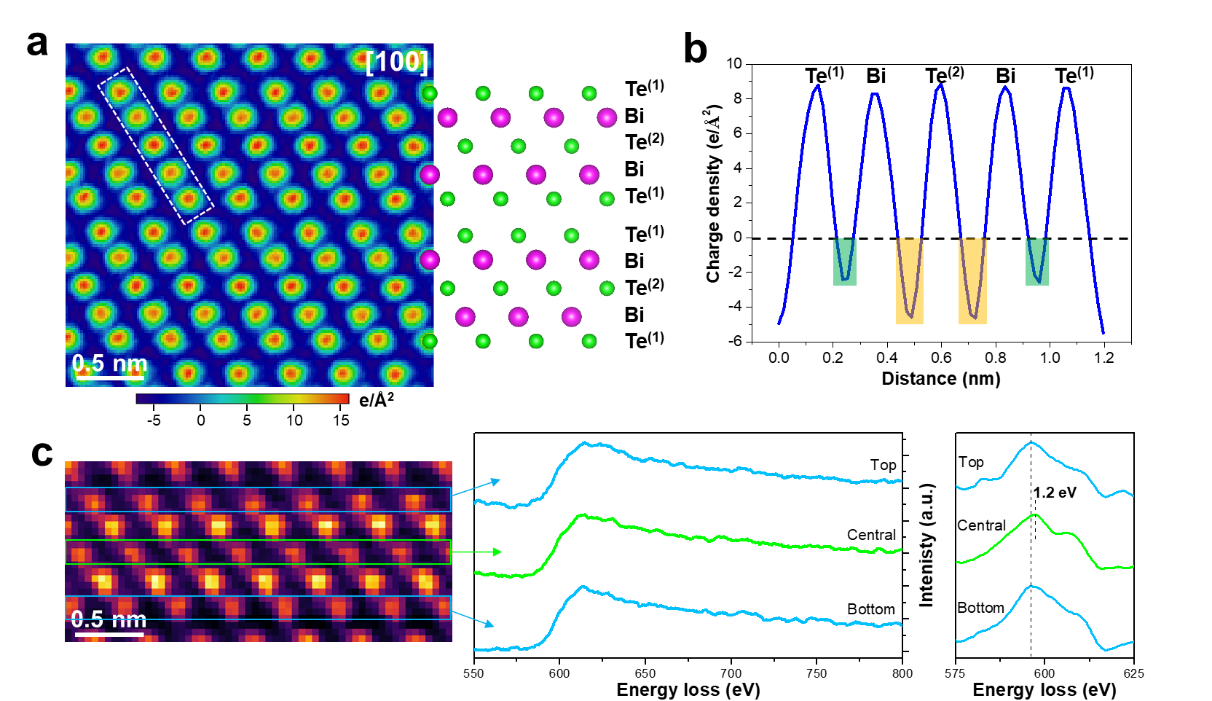
Figure 2. Atomic-scale analysis of bonding disparity in Bi2Te3.
The study reveals that this atomic-scale chemical inhomogeneity directly governs thermoelectric properties. As the degree of Se enrichment in the central layer varies, the bonding polarity, band gap, and Seebeck coefficient evolve in a correlated manner (Fig. 3); larger differences in Se concentration between the central and outer layers lead to stronger bonding polarity, a wider band gap, and a higher Seebeck coefficient. Based on these relationships, the researchers established the first quantitative model linking atomic-scale site occupancy with macroscopic thermoelectric transport properties, enabling cross-scale prediction from electron microscopy observations to macroscopic transport parameters.
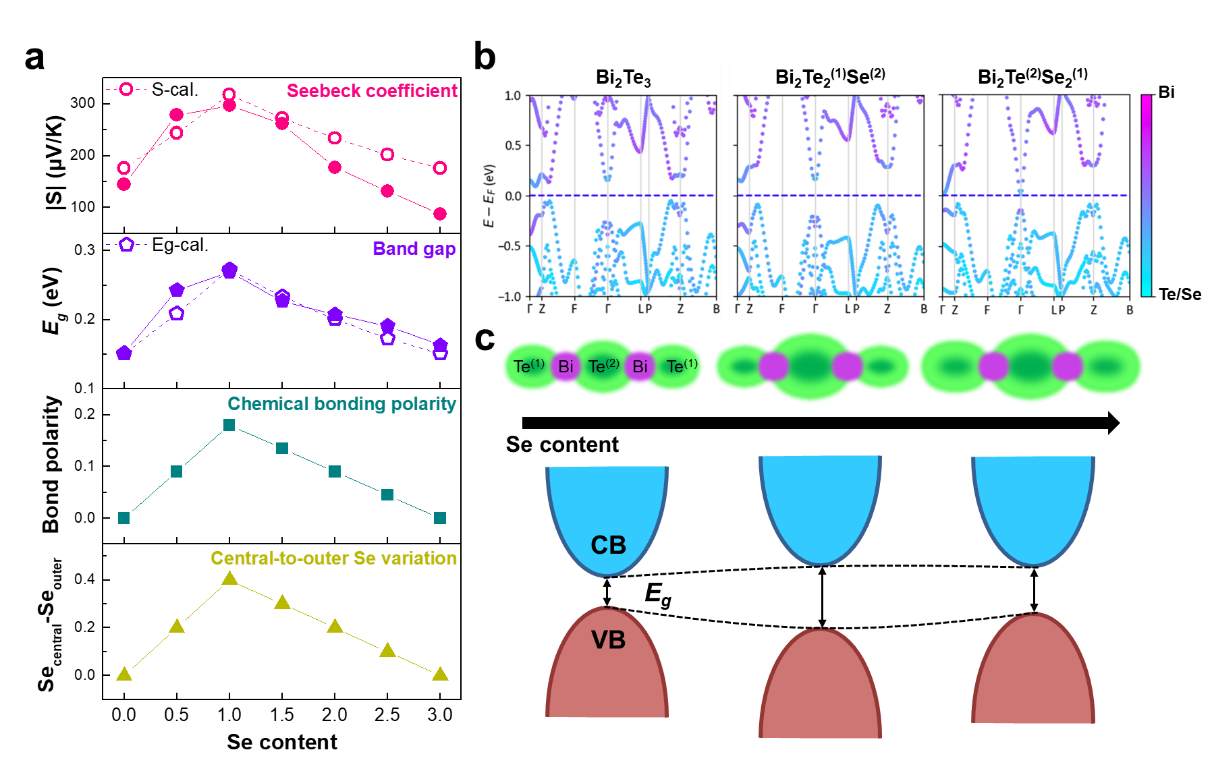
Figure 3. Quantitative relationship between local chemical inhomogeneity and electronic transport properties in Bi2Te3-xSex.
This work establishes a comprehensive cross-scale mechanism connecting atomic structure, chemical bonding, electronic structure, and transport properties in Bi2Te3-based thermoelectric materials. The findings not only provide new theoretical guidance for the design of high-performance thermoelectric materials, but also offer an important paradigm for understanding structure–property coupling in layered functional materials.
Dr. Wu WANG, Research Assistant Professor at SUSTech (now Associate Professor at Shenzhen Technology University), Dr. Juan CUI from the Institute of Applied Physics and Computational Mathematics, and PhD student Zhongbin WANG from the Department of Physics at SUSTech are co-first authors of the paper. Dr. Wu WANG and Professor Jiaqing HE are the corresponding authors. SUSTech is the lead institution of the study.
Paper Link: https://link.aps.org/doi/10.1103/qqrq-ths7
Proofread ByNoah Crockett, Junxi KE
Photo ByYan QIU