A collaborative team led by Assistant Professor Xin HONG of the Institute for Homeostatic Medicine, the Department of Biochemistry at the Southern University of Science and Technology (SUSTech), and the School of Medicine at SUSTech, in collaboration with several national clinical centers across China, published a research article in the prestigious journal Cancer Discovery entitled “GPNMB Drives Brain Metastasis by Sculpting a Pathological Endothelial-Immune Interactome.” Focusing on the central question of how circulating tumor cells (CTCs) breach the blood-brain barrier (BBB) and reshape the immune microenvironment of the brain, the study employs multi-omic strategies, including single-cell transcriptomics of CTCs, spatial transcriptomics of brain metastases, whole-exome sequencing, plasma proteomics, in vitro and in vivo functional assays, and clinical validation in independent clinical cohorts. The work identifies the CTC-derived secreted factor GPNMB as a driver of BBB disruption, increased endothelial permeability, and subsequent immune cell infiltration through the CXCL12-CXCR4 axis, culminating in an immunosuppressive brain metastases niche. Importantly, the study reveals CBX3+GPNMB+CTCs and plasma CXCL12 as potential non-invasive liquid biopsy biomarkers for patients with brain metastases, and suggests that combined blockade of GPNMB and PD-1 may offer a rational strategy for precision immunotherapy in this setting.

Brain metastasis is among the most devastating manifestations of advanced solid malignancy and occurs frequently in patients with lung cancer, breast cancer, melanoma, colorectal cancer, and renal cancer. Because of the specialized architecture of the BBB, extracranial tumor cells must undergo a highly coordinated process of vascular adhesion, endothelial engagement, and extravasation before they can colonize the brain parenchyma. CTCs are widely considered the “seeds” of haematogenous metastasis; however, only a rare subset can survive circulation, adhere to the cerebral microvasculature, compromise BBB integrity, and ultimately establish intracranial lesions. How these CTCs communicate with brain endothelial cells, and how this interaction subsequently remodels the immune microenvironment of brain metastases, has remained incompletely understood. This study addresses this continuum from the earliest vascular entry step to later immune remodeling by building an integrated mechanistic framework centred on the CTC-BBB-immune microenvironment axis.
The investigators first established a multi-omic platform to profile patient-derived CTCs and brain metastatic lesions. In total, the study analysed 63 samples from 60 individuals, including an in-house cohort of 10 patients with brain metastases. Whole-exome sequencing was performed on selected metastatic brain tissues and matched peripheral blood mononuclear cells; high-resolution spatial transcriptomics were applied to four metastatic brain specimens; and bulk RNA-seq validation was conducted in an additional cohort of 29 metastatic brain samples. Using an advanced microfluidic CTC-isolation platform, the team captured 79 CTCs from the peripheral blood of seven patients with brain metastases and subjected them to a Smart-seq3 protocol optimized for low-viability CTCs. Plasma samples from 15 patients with lung cancer brain metastases were further analyzed by Olink proteomics. This design enabled cross-validation across metastatic seeds in the blood, tissue architecture in intracranial lesions, circulating protein biomarkers, and functional experimental systems (Fig. 1). The authors found that CTCs from patients with brain metastases did not express the classical EPCAM-high epithelial CTC phenotype but instead displayed non-canonical transcriptomic signatures. Further analysis resolved two major CTC subsets: one with myeloid-like transcriptional programs and another with erythroid/platelet-associated features (Fig. 1B, 1C). Although epithelial markers were expressed at low levels at the mRNA level, somatic mutation matching, copy-number-variation inference, and multiplex immunofluorescence confirmed their malignancy (Fig. 1D, 1E). These findings suggest that brain metastasis-associated CTCs may have acquired haematopoietic traits that enable immune evasion and adaptation to the circulatory and cerebral vascular microenvironments.
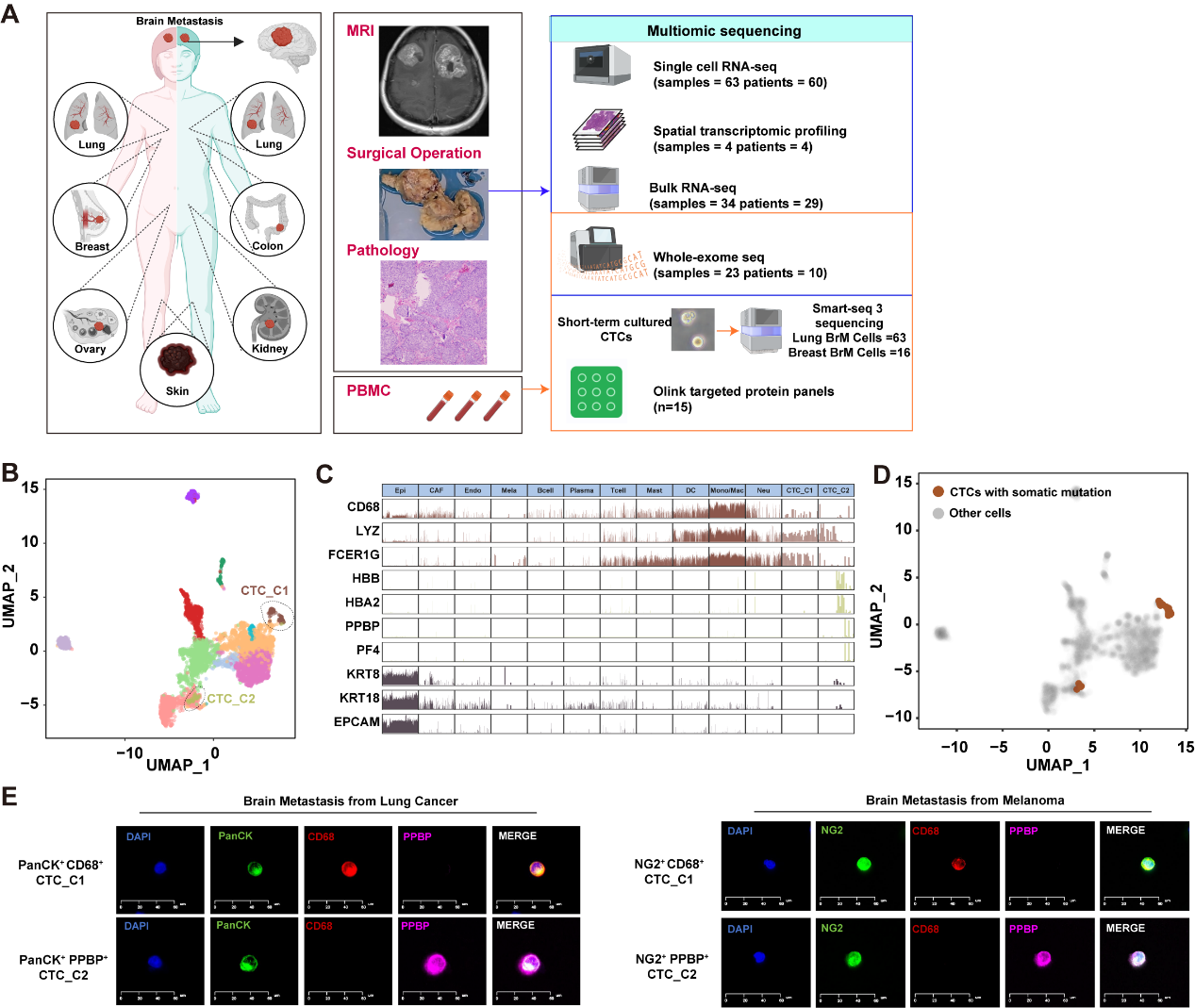
Figure 1. Single-cell multi-omic characterization of circulating tumor cells (CTCs) from patients with brain metastases.
By intersecting genes highly expressed in CTCs with the secretome of an in vitro BBB model, the investigators identified GPNMB as a secreted protein enriched in brain-metastasis-associated CTCs and potentially involved in BBB disruption (Fig. 2A). Functional assays showed that co-culture of brain endothelial cells with GPNMB-overexpressing H1975 lung cancer cells or A375 melanoma cells markedly impaired endothelial tube formation and reduced transendothelial electrical resistance (TEER). These endothelial defects were substantially reduced by treatment with a GPNMB-targeting antibody (Fig. 2B, 2C). Thus, CTC-derived GPNMB is not merely a correlative marker but a functional mediator of increased BBB permeability and brain endothelial dysfunction. In vivo, the team established multiple brain metastasis models and further demonstrated that GPNMB promotes intracranial colonization. In a B16 melanoma brain metastasis model, GPNMB overexpression shortened mouse survival and increased both systemic and brain metastatic burden, whereas “in vivo“ si-GPNMB treatment prolonged survival and reduced intracranial metastasis (Fig. 2D-G). These results indicate that GPNMB exerts a conserved pro-metastatic function across distinct tumor models by perturbing BBB integrity and facilitating brain colonization.

Figure 2. CTC-secreted GPNMB drives brain endothelial dysfunction and metastatic progression.
The study delineates how GPNMB acts on brain endothelial cells. Ligand-receptor interaction and protein-protein interaction prediction analyses suggested that GPNMB may interact with EGFR on brain endothelial cells (Fig. 3A), and co-immunoprecipitation confirmed a physical association between GPNMB and EGFR (Fig. 3B). Subsequent experiments showed that GPNMB stimulation decreases EGFR protein stability and promotes CBL-mediated EGFR ubiquitination and degradation (Fig. 3C, 3D). The authors further found that the role of EGFR in maintaining BBB integrity is closely linked to the m6A epitranscriptomic pathway. The m6A demethylase FTO was markedly reduced in endothelial cells from brain metastases (Fig. 3E), and EGFR knockdown decreased FTO expression while increasing global m6A methylation levels (Fig. 3F, 3G). MeRIP-seq and RIP assays demonstrated that FTO directly modulates m6A modification of TJP1 mRNA and maintains its stability (Fig. 3H, 3I). The m6A reader YTHDF2, in turn, binds TJP1 mRNA and promotes its degradation (Fig. 3J). YTHDF2 knockdown rescued the reduction in TJP1 expression and the endothelial dysfunction caused by FTO loss (Fig. 3K, 3L). Together, these data define a GPNMB-EGFR-FTO-YTHDF2-TJP1 axis that explains how CTC-secreted GPNMB disrupts the BBB through an RNA epitranscriptomic mechanism.

Figure 3. GPNMB promotes brain vascular dysfunction by suppressing the EGFR-FTO-TJP1 axis.
Beyond its role in BBB disruption, a key conceptual advance of this study is that GPNMB does not simply help tumor cells “enter” the brain; it also induces an immunosuppressive microenvironment of brain metastases. In bulk RNA-seq datasets from brain metastatic samples, GPNMB-associated signatures correlated positively with T-cell infiltration, effector T-cell programs, and exhausted T-cell signatures (Fig. 4A). High-resolution spatial transcriptomics of four brain metastatic specimens revealed that PECAM1+ endothelial cells were spatially adjacent to immune cells associated with tertiary lymphoid structures, including T cells, B cells, and plasma cells (Fig. 4B). Multiplex immunohistochemistry further confirmed immune-cell accumulation around abnormal vascular structures within brain metastatic lesions (Fig. 4C, 4D). These findings suggest that abnormal brain endothelial cells are not passive barrier components but active organizers of immune-cell recruitment and local immune remodeling. Ligand-receptor analysis further highlighted sustained enrichment of the CXCL12-CXCR4 signaling axis in brain metastases (Fig. 4E). In vitro assays demonstrated that GPNMB treatment stimulates CXCL12 secretion by brain endothelial cells. Treatment with the anti-GPNMB antibody CDX-011 or the CXCL12 inhibitor NOX-A12 significantly reduced the capacity of endothelial cells to recruit CD3+ T cells and CD19+ B cells, and also suppressed recruitment of CXCR4+ immune cells (Fig. 4F). In plasma samples from patients with brain metastases, CXCL12 protein levels were significantly higher in patients with high immune infiltration than in those with low immune infiltration, supporting plasma CXCL12 as a potential non-invasive biomarker of the immune-infiltrated state of brain metastases (Fig. 4G).

Figure 4. Abnormal endothelial cells in brain metastases promote immune-cell infiltration through the CXCL12-CXCR4 axis.
The team evaluated the therapeutic potential of combined GPNMB targeting and PD-1 immune checkpoint blockade in brain metastasis. In an LLC mouse lung cancer brain metastasis model, GPNMB overexpression markedly increased systemic tumor burden and brain metastatic outgrowth, whereas in vivo silencing of GPNMB significantly suppressed brain metastasis. Notably, dual therapy with anti-PD-1 antibody and Si-GPNMB further enhanced the anti-metastatic effect compared with the GPNMB overexpressing group or T1 monotherapy (Fig. 5A-F). Compared with the negative control group, mice in the GPNMB overexpressing group showed increased total CD31+ endothelial cells in the brain but a reduced proportion of CD31+CDH5+ endothelial cells, indicating severe disruption of the cerebral microvasculature (Fig. 5G, 5H). GPNMB inhibition restored endothelial integrity in brain tissue and reduced CD8+ T-cell infiltration when the NC, OE, and T1 groups were compared (Fig. 5I-K).

Figure 5. The LLC in vivo brain metastasis model supports the therapeutic efficacy of combined GPNMB and PD-1 blockade.
This study reveals a molecular regulon by which CTCs secrete GPNMB to disrupt the BBB and remodel the immune microenvironment of brain metastases. CTC-derived GPNMB binds EGFR on brain endothelial cells, triggers CBL-mediated EGFR ubiquitination and degradation, suppresses FTO expression, and disrupts endothelial tight junctions through YTHDF2-dependent m6A regulation of TJP1. In parallel, GPNMB-educated abnormal brain endothelial cells promote immune cell infiltration into the brain metastatic microenvironment through the CXCL12-CXCR4 axis, and establish a pathological vascular-immune interactome with immunosuppressive hallmark signatures. The work provides a conceptual framework for understanding how CTCs traverse the BBB and initiate brain colonization. It also identifies CBX3+GPNMB+ CTCs and plasma CXCL12 as candidate liquid-biopsy markers for patient stratification and prediction of immunotherapeutic response. The enhanced anti-metastatic activity of combined GPNMB and PD-1 blockade in animal models offers a new rationale for precision immunotherapy in patients with brain metastases.

Working Model.
The study was conducted in collaboration with the neurosurgery teams of Professors Qing LIU and Jun TAN at the Xiangya School of Medicine of the Central South University; Professor Feiqiu WEN’s team at Shenzhen Children’s Hospital; the teams of Chunying LI and Weinan GUO at Xijing Hospital, Air Force Medical University; and the nasopharyngeal carcinoma team led by Sailan LIU at Sun Yat-sen University Cancer Center. The co-first authors of this paper are doctoral graduate student Xuefei LIU from SUSTech (Class of 2022), Dr. Jun TAN from the Department of Neurosurgery of the Xiangya School of Medicine of the Central South University; Chun WU, a doctoral graduate student enrolled in 2023 at Sun Yat-sen University Cancer Center and a visiting student at SUSTech, master graduate student Guanyin HUANG from SUSTech (Class of 2022), and Yixin CHENG, a doctoral graduate student enrolled in 2024 at Sun Yat-sen University Cancer Center and a visiting student at SUSTech. Assistant Professor Xin HONG from the Department of Biochemistry of the SUSTech Institute of Homeostatic Medicine at the School of Medicine at SUSTech; Professor Feiqiu WEN from the Shenzhen Children’s Hospital; Professor Qing LIU from the Department of Neurosurgery of Xiangya School of Medicine at Central South University; Professor Sailan LIU from the Department of Nasopharyngeal Carcinoma of Sun Yat-sen University Cancer Center; and Professors Chunying LI and Weinan GUO from Xijing Hospital of the Air Force Medical University, are co-corresponding authors. SUSTech serves as the first affiliation and the last corresponding affiliation.
Article Link: https://pubmed.ncbi.nlm.nih.gov/41973996/
(DOI: 10.1158/2159-8290.CD-25-1663)
Proofread ByNoah Crockett, Junxi KE
Photo ByYan QIU