Diabetes mellitus (DM) is a chronic metabolic disorder affecting over 500 million individuals worldwide. It is characterized by impaired insulin secretion and/or peripheral insulin resistance, leading to hyperglycemia and a spectrum of associated complications. In recent years, islet transplantation has emerged as a promising therapeutic approach for diabetes, aiming to restore endogenous insulin production and maintain glucose homeostasis.
Although islet transplantation offers improved metabolic outcomes and a reduced incidence of severe hypoglycemia compared to conventional insulin therapy, its clinical application remains limited by several challenges, including a scarcity of donor tissues and the diminished insulin secretory capacity of isolated islets. A deeper understanding of the regulatory mechanisms governing β-cell function may provide new therapeutic avenues for diabetes management and islet transplantation.
Associate Professor Huiling Cao’s team from the Department of Biochemistry and the Institute of Biomedical Sciences at the Southern University of Science and Technology (SUSTech) has identified March5 and Trim28 as novel and critical regulators of pancreatic β-cell function. Their findings offer a promising therapeutic strategy for protecting and restoring β-cell functionality in diabetes.
Their study, entitled “March5-mediated Trim28 degradation preserves islet β-cell function in mice,” has been published in Nature Communications.
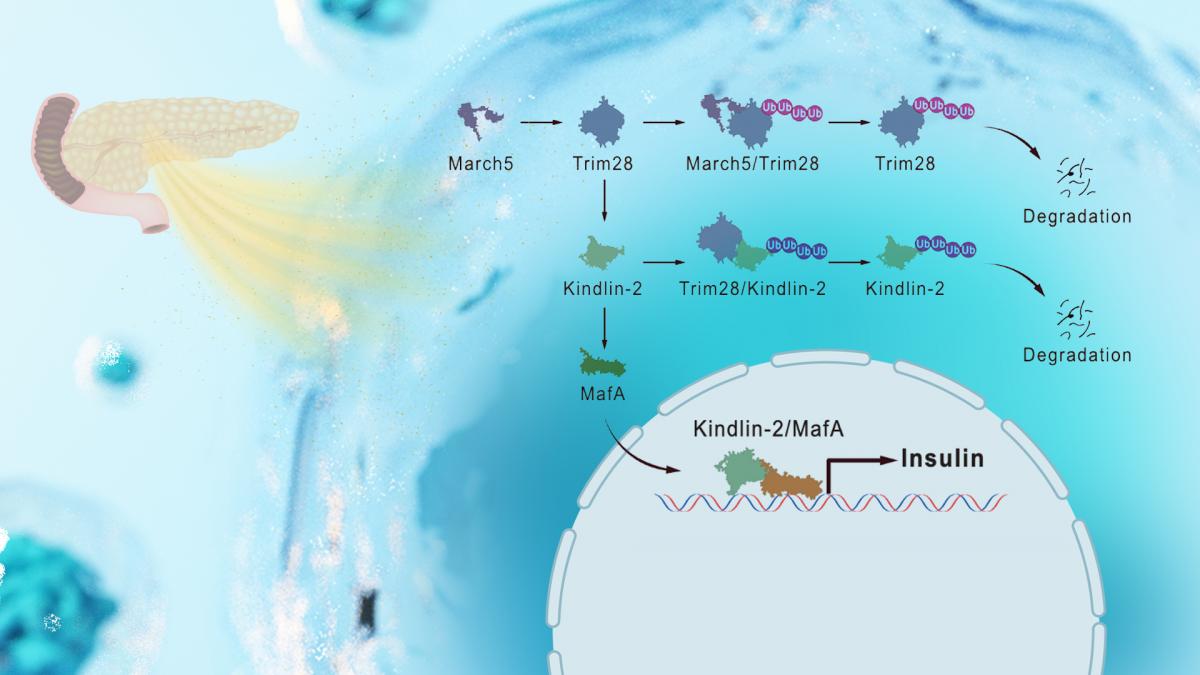
The researchers first employed β-cell–specific knockout and knock-in mouse models to manipulate March5 expression in pancreatic β-cells. The results demonstrated that overexpression of March5 significantly enhanced insulin secretion under both normal chow and high-fat diet conditions, without affecting β-cell differentiation or mass. Further mechanistic investigations revealed that the loss of March5 impaired islet function through suppression of Kindlin-2, a known key regulator of pancreatic β-cell activity. This was confirmed in double-heterozygous knockout mice, supporting the functional interplay between March5 and Kindlin-2.
Moreover, β-cell–specific deletion of Trim28 in March5-deficient mice effectively improved glucose homeostasis, rescuing the impaired glucose tolerance and reduced glucose-stimulated insulin secretion (GSIS). These findings indicate that March5 maintains β-cell function by promoting Trim28 ubiquitination and degradation.
Subsequent experiments showed that Trim28 interacts directly with Kindlin-2 and modulates its ubiquitination, thereby influencing MafA activity and β-cell function. Mechanistically, this highlights a regulatory axis wherein Trim28 governs MafA-mediated transcriptional control through Kindlin-2 stabilization.
Finally, islet transplantation from mice with β-cell–specific March5 overexpression or Trim28 deficiency markedly improved glycemic control in streptozotocin (STZ)-induced diabetic mouse models. In ex vivo assays, upregulation of MARCH5 or downregulation of TRIM28 enhanced insulin production and secretion in isolated islets.
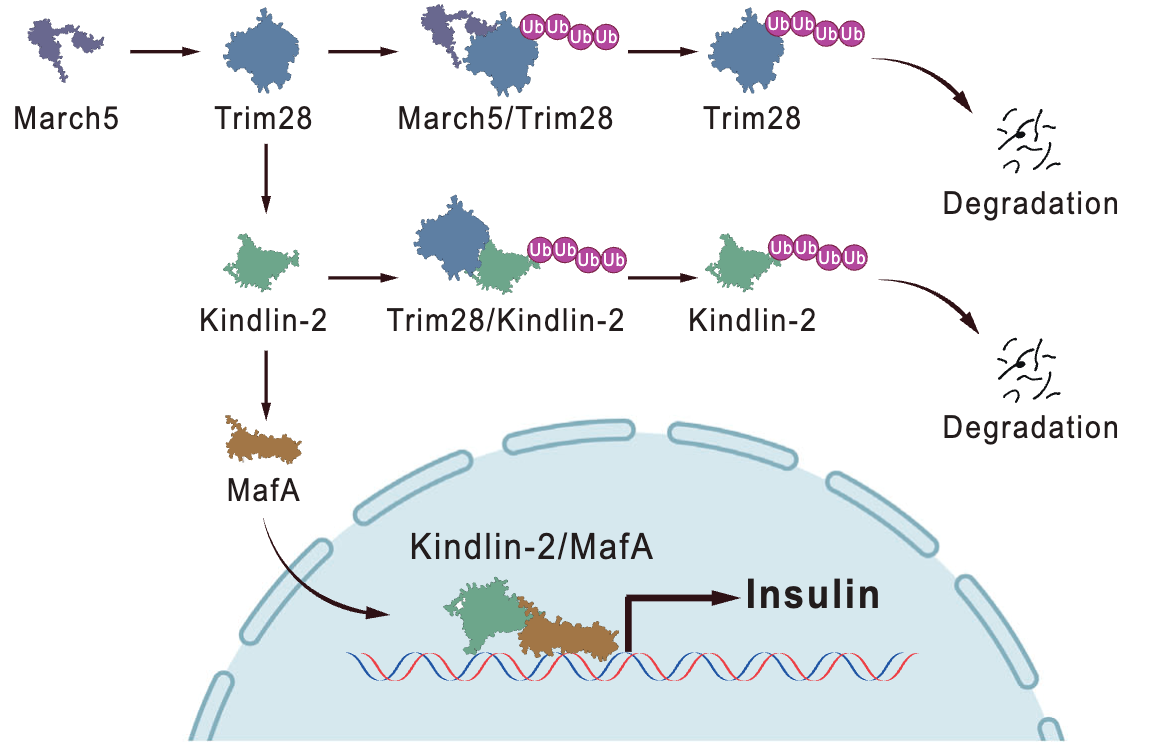
Figure 1. Molecular mechanism by which the March5/Trim28/Kindlin-2/MafA axis regulates insulin expression in pancreatic β-cells
This study uncovers a critical role for the March5/Trim28/Kindlin-2/MafA axis in regulating insulin expression and secretion in pancreatic β-cells, and suggests this signaling cascade as a promising therapeutic target for restoring β-cell function in diabetes.
Postdoctoral fellow Yangshan Chen, master’s student Wei Pang, and Ph.D. candidate Guixing Ma are co-first authors of this work. Associate Professor Huiling Cao is the corresponding author.
Paper link: https://www.nature.com/articles/s41467-025-62587-z
To read all stories about SUSTech science, subscribe to the monthly SUSTech Newsletter.
Proofread ByAdrian Cremin, Yuwen ZENG
Photo ByDepartment of Biochemistry